Uranium
 Symbol of uranium that would be seem on the periodic table
Symbol of uranium that would be seem on the periodic table
Uranium's symbol is U
Atomic number is 238.02891
Uranium is a metal and is a solid at room temperature
Density is 18.95 grams per cubic centimeter
Uranium melts at 1408 K
Uranium boils at 4404 K
Molar mass is 238.02891
Atomic radius is 175 pm
Uranium's family on the periodic table is Actinides
Uranium's period is 7
Uranium's block is f
6 valence electrons
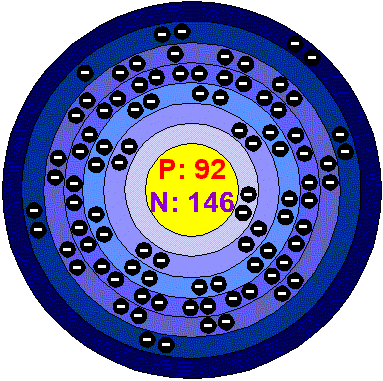
Electron configuration long form 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2 5f 4
Electron configuration short form [Rn] 7s2 5f 4
Four quantum numbers for last electron in orbital diagram n=5 L=3 ML=-3,-2,-1,0,1,2,3 MS= +1/2
Atomic number is 238.02891
Uranium is a metal and is a solid at room temperature
Density is 18.95 grams per cubic centimeter
Uranium melts at 1408 K
Uranium boils at 4404 K
Molar mass is 238.02891
Atomic radius is 175 pm
Uranium's family on the periodic table is Actinides
Uranium's period is 7
Uranium's block is f
6 valence electrons
Electron configuration long form 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2 5f 4
Electron configuration short form [Rn] 7s2 5f 4
Four quantum numbers for last electron in orbital diagram n=5 L=3 ML=-3,-2,-1,0,1,2,3 MS= +1/2